
Other comorbidities have been described in patients who developed NSF, including acute pro-inflammatory states, metabolic acidosis, increased iron, calcium or phosphate levels, immunosuppression, vasculopathy, high dose erythropoietin therapy and infection 3. Many of the reported cases of NSF have been in patients before or after liver transplant. Considering this clarification, FDA has not received reports of NSF among patients with normal renal function or moderate renal insufficiency”. Additional details have clarified that the patients actually were in acute renal failure at the time they received a gadolinium based contrast agent. Since issuing the information in December 2006, FDA has received new information regarding these patients. In 2009, the FDA determined that moderate renal impairment (eGFR of 30-60) was NOT a risk factor for NSF, reversing the position it had taken in December 2006, and stated 2 “The December information was based upon reports of NSF among patients with purportedly moderate renal insufficiency. 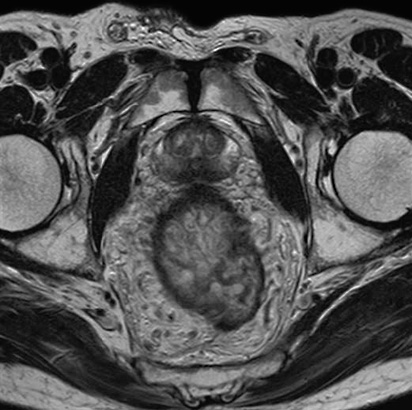
The exact etiology of NSF is unclear but most reported cases have been documented in patients with severe acute or chronic renal failure, with a glomerular filtration rate (GFR) 30 1. NSF is generally seen in the middle-aged, but has also been reported in the elderly and in children. This rare but serious systemic disease is characterized by fibrosis of the skin and other tissues throughout the body. This changed in 2006 when the FDA first reported the association between nephrogenic systemic fibrosis (NSF) and intravenous gadolinium administration. See /license for the full LOINC copyright and license.The administration of intravenous gadolinium-containing contrast media was historically considered safe in patients with impaired renal function. 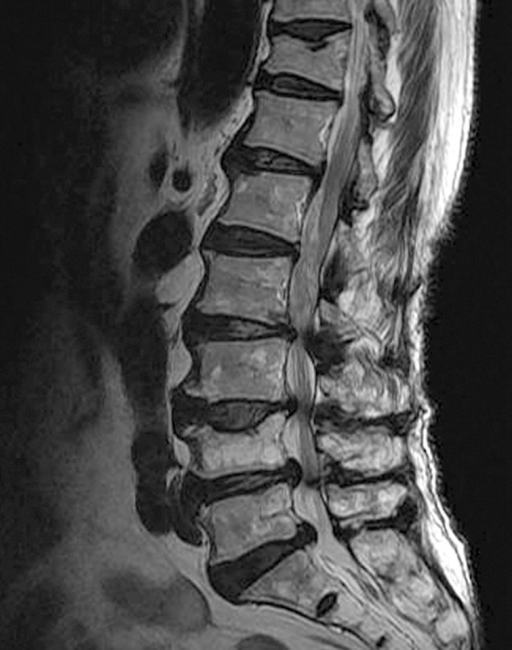
and the Logical Observation Identifiers Names and Codes (LOINC) Committee. To the extent included herein, the LOINC table and LOINC codes are copyright © 1995-2022, Regenstrief Institute, Inc. LOINC FHIR ® API Example - CodeSystem Request Get Info $lookup?system= LOINC CopyrightĬopyright © 2022 Regenstrief Institute, Inc. Observation Both Language Variants Get Info zh-CNChinese (China) 多层^采用与不采用静脉注射对比剂: 发现: 时间点: 颞骨: 叙述型: MRI it-ITItalian (Italy) Sezioni multiple^Con & senza contrasto EV: Osservazione: Pt: Osso temporale: Nar: MRI pt-BRPortuguese (Brazil) Multi cortes^com e sem contraste EV: Achado: Pt: Ossos temporais: Nar: RM es-ARSpanish (Argentina) : hallazgo: punto en el tiempo: huesos faciales+maxilar: Narrativo: resonancia magnética es-MXSpanish (Mexico) Contraste multisección ^ W & WO IV: Tipo: Punto temporal: Hueso temporal: Narrativo: MRI Related Names Version 2.73 36836-5 Deprecated Temporal bones MRI W & WO contrast IV Deprecated Status Information Status Deprecated Reason Duplicate Comment Temporal bone MRI would include brain Map-Toįully-Specified Name Component Multisection^W & WO contrast IV Property Find Time Pt System Temporal bone Scale Nar Method MRI Additional Names Short Name Deprecated Temporal bones MRI W+WO contr Basic Attributes Class RAD Type Clinical First Released Version 2.14 Last Updated Version 2.38 Order vs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
